Verdict
Jurisdiction and Limitation for Adjudication
In the Supreme Court, Criminal Appellate
Criminal Appeal No. of 2026
[arising out of SLP (Criminal) No.4524 of 2023]
State of Kerala …………… Appellants
A1: The State of Kerala represented by Home Secretary
A2: The drugs inspector (intelligence branch) office of
Assistant drugs controller, Thrissur, Kerala
Vs
M/s Panacea Biotec Ltd & Another ………………Respondents
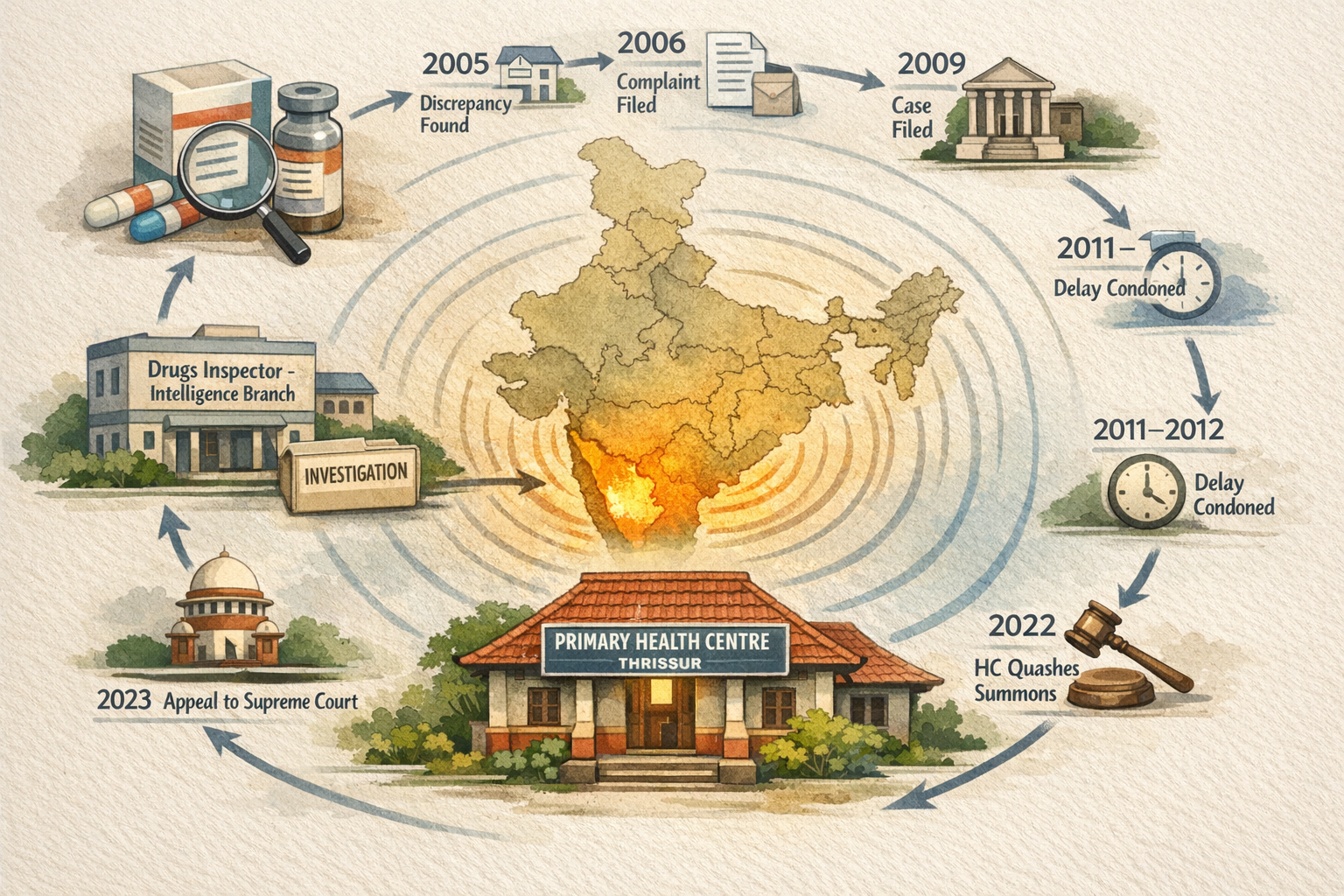
Facts
- In October 2005, the Primary Health Centre (“PHC”) in Thrissur noticed a discrepancy in nomenclature of a medicinal drug as given on the labels on the carton, body and seal cap of the drug vial. The discrepancy pertained to the very composition of the drug that would mislead the user to believe it to be a vaccine for Hepatitis. The PHC brought it to the notice of Mr. Joy Mandi, the retail supplier (‘supplier’). In January 2006, the supplier in turn lodged a complaint with the office of Drugs Inspector (“Intelligence Branch”), Thrissur against the manufacturers and wholesale suppliers of the drug (“Panacea Biotec”)
- Though the intelligence branch commenced enquiry into the complaint immediately, it was only in January 2009, 3 years after the complaint, it filed a case with the Chief Judicial Magistrate, Thrissur [“CJM”] for offence under Drugs and Cosmetics Act, 1940 [“Act”] and Drugs and Cosmetics Rules, 1945 [“Rules”].
- In August 2009, Panacea Biotec filed a criminal case in the Kerala High Court, under Section 482 of Code of Criminal Procedure [“Code”], seeking the quashing of CJM’s Summons.
- In June 2011, the High Court directed CJM to consider whether the delay on part of Panacea Biotec in filing the case with CJM could be condoned in terms of Section 473 of the Code.
- In July 2012, the CJM ordered condoning the delay under Section 473 and again summoned Panacea Biotec.
- In August 2012, Panacea Biotec filed another criminal case in the Kerala High Court under Sections 482 (inherent powers of HC) and 202 (mandatory inquiry for sufficiency of grounds for complaint) and once again sought the quashing of the CJM’s Summons.
- In July 2022, the Kerala High Court quashed the CJM’s Summons on grounds that though Panacea Biotec resided outside his jurisdiction, the CJM failed to conduct inquiry mandated under Section 202. The High Court, however, did not interfere with CJM’s July 2012 Order condoning the delay under Section 473. Effectively, the HC did not allow Panacea Biotec from even being summoned to court for an inquiry into the complaint.
- In 2023, Panacea Biotec filed Special Leave Petition (Criminal) No. 4524 of 2023, seeking the setting aside of High Court order that quashed the CJM Summons.
Issues involved
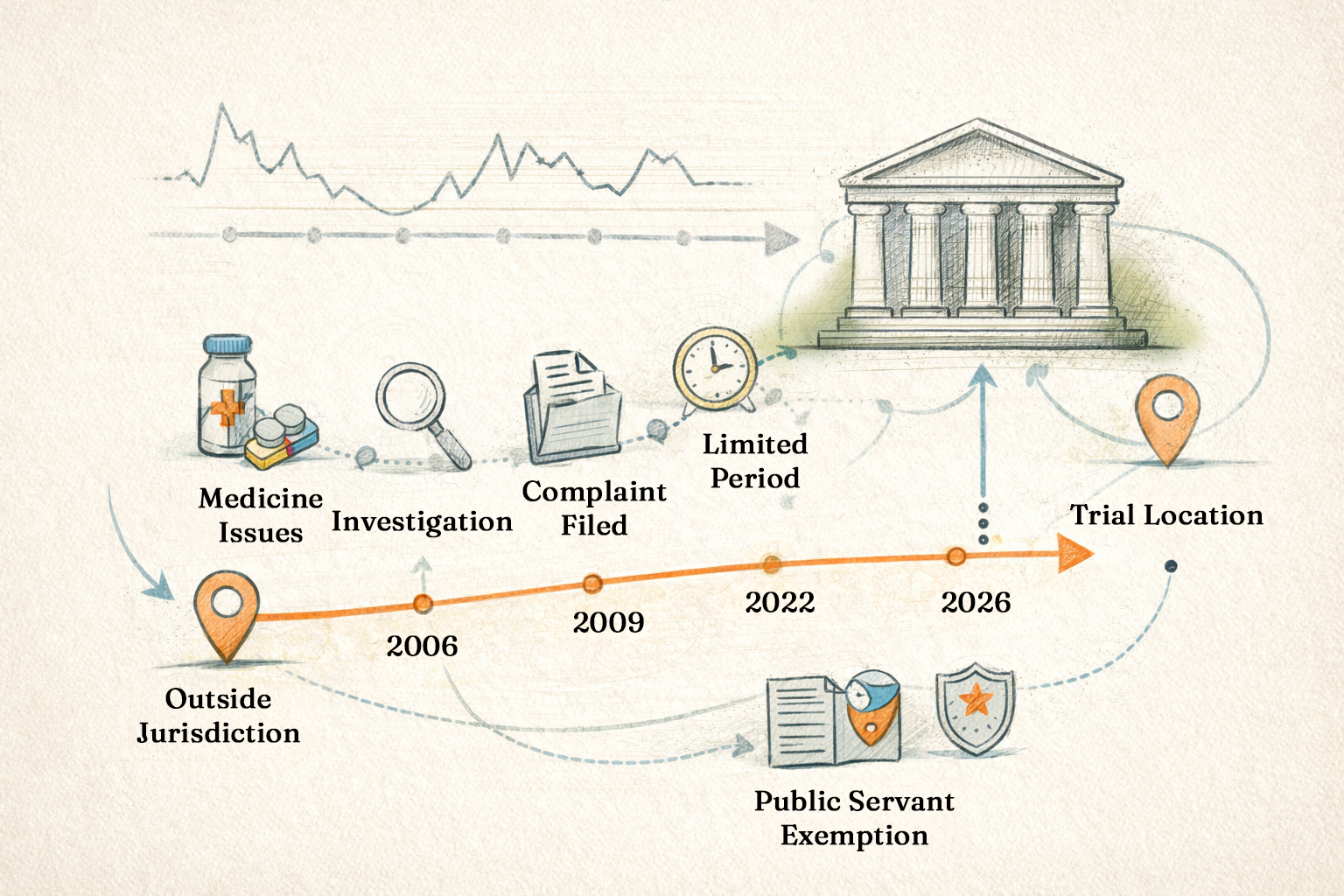
- What are the grounds for and the extent of ‘time limitation’ applicable to a Court for taking cognisance of a complaint filed before it?
- What is the nature of ‘territorial jurisdiction’ applicable to a Court for adjudicating on such complaints?
- Will it make any difference in such cases if the complainant is a ‘public servant’?
Reference Cases
Supreme Court’s verdicts in following cases have been referred to in the adjudication of this case:
- Cheminova India Limited v/s State of Punjab, 2021 SCC OnLine SC 5736
- Birla Corporation Limited v/s Adventz Investments and Holdings Limited, 2019 16 SCC 610
Analysis
a) Grounds for and extent of ‘time limitation’ applicable to a Court for taking cognisance of a complaint:
- While considering plea of Panacea Biotec, the Kerala HC relied primarily on Sections 468 and 469 of Code. Section 469 specifies the limitation period (6 months to 3 years) for court to take cognisance of complaint and Section 469 specifies the date of commencement of this limitation period. The Kerala HC was convinced that intelligence branch approached CJM only after expiry of the limitation period in the present case.
- In reality, the intelligence branch conducted a long-drawn inquiry to trace the entire sequence of commercial transactions involved in supply of the drug and collecting documentary evidence for the complaint. It was only after conclusion of the inquiry that it was in a position to identify all the persons who could be held responsible for the offence of ‘wrong labelling’. It was only in April 2006 that the identities of all the alleged offenders in this case could be established. Clause 1 (c) of Section 469 clearly specifies that the limitation period commences from the date on which the ‘identity of the offender becomes known to the person aggrieved by the offence’. As such, the 3 -year limitation period commences from April 2006 only; the complaint with CJM had been lodged in January 2009, which is within the deadline of April 2009.
- The nature of the alleged offence namely, wrong labelling, is such that it not only mislead the sellers but also endangered the lives of potential users of the drug which was labelled as a particular vaccine. Section 473 permits any court to take cognisance of an offence even after expiry of limitation period notwithstanding anything contained in any other provision in the Code, provided (i) court is satisfied by the explanation given for delay in filing complaint, or (ii) court finds it necessary to take cognisance of the complaint in the interest of justice.
Based on the above analysis, the Hon’ble SC overruled the Kerala HC order quashing the summons to Panacea Biotec.
b) Nature of ‘territorial jurisdiction’ applicable to a Court for adjudicating on a complaint:
- Panacea Biotec argued in the HC that they being located in Delhi are outside the jurisdiction of Thrissur CJM and that under Section 202 of the Act, the CJM cannot receive complaint (i) without examination of complainant and witnesses under oath and (ii) without investigation into complaint by a police officer or any person deemed fit, for purpose of deciding if sufficient grounds exist for the complaint. Accepting this argument, HC quashed the CJM summons to Panacea Biotec.
- The drugs inspector (intelligence branch) the complainant in this case, is a Gazetted officer authorised by the state govt to specifically investigate into such offences. In other words, it is on behalf of the state govt that the complaint has been lodged with CJM; such complaints do not require to be taken under oath. Moreover, he is a ‘public servant’ discharging his official duty.
Section 200 of Code specifically empowers a public servant discharging his official duty to make the complaint which is exempted from examination under oath. This is an exemption that is not available to a private complainant.
- Recognising the Drug inspector (intelligence branch) as ‘public servant discharging his duty’, the SC recognised his complaint to the CJM and accordingly set aside the Kerala High Court’s order quashing the summons to Panacea Biotec.
For full text of the judgement:
https://api.sci.gov.in/supremecourt/2023/5043/5043_2023_13_1501_69014_Judgement_26-Feb-2026.pdf